MEDAPRINT is a concentrated disinfectant intended for the cleaning and disinfection of dental impressions prior to handling, transport, or laboratory processing. It is designed to support infection control procedures in dental practices and to reduce the risk of cross-contamination between the clinic and the laboratory.
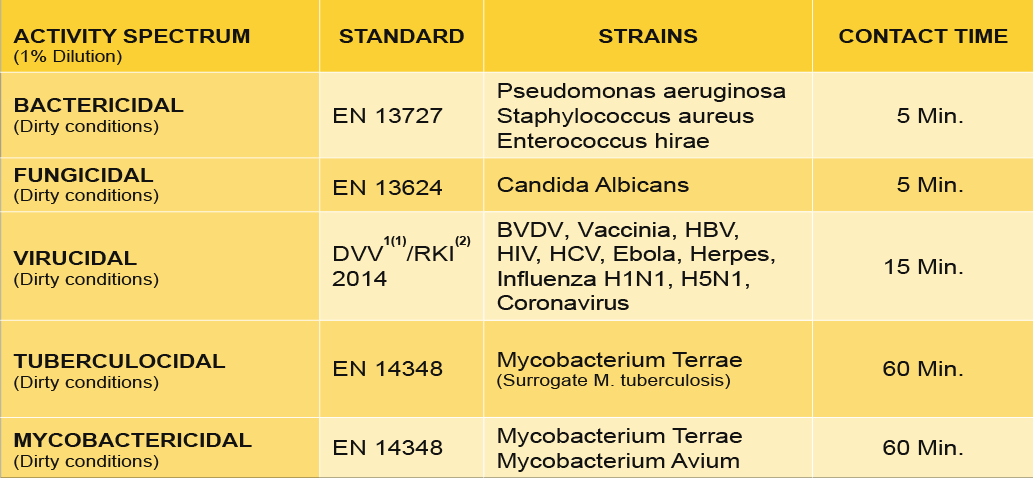
The formulation delivers a broad antimicrobial spectrum, including bactericidal, fungicidal, virucidal, tuberculocidal, and mycobactericidal activity, ensuring reliable reduction of cross-contamination risks between the dental clinic and the laboratory environment.
MEDAPRINT is fully compatible with all commonly used impression materials, including alginates, silicones, polyethers, and hydrocolloids, without affecting dimensional stability, surface detail or accuracy.
The product is intended for use in an immersion bath, ensuring complete contact between the disinfectant solution and all impression surfaces. This supports consistent and reliable disinfection results as part of routine dental impression reprocessing.
Properties
Microbiological efficacy
* Including all antibiotic resistant bacteria such as Escherichia coli, Klebsiella pneumoniae, Streptococcus pneumoniae, etc.)
(1) DVV: Deutsche Vereinigung zur Bekämpfung der Viruskrankheiten / German Association for the Control of Virus Diseases
(2) RKI: Robert Koch Institute – German Federal Health Authority
Compatibility
MEDAPRINT is compatible with impressions from alginates, silicones, polyethers and hydrocolloids.
MEDAPRINT is not compatible with disinfecting preparations containing aldehydes.
Packaging
Physical properties
Composition
N- (3-aminopropyl) -N dodecylpropano-1,3-diamine, non-ionic surfactants <5%, isopropanol, excipients.