MEDAPRINT FOAM is a ready-to-use foaming disinfectant spray developed for the rapid cleaning and disinfection of dental impressions immediately after removal from the oral cavity. It offers a practical, chairside-friendly alternative to immersion disinfection for dental practices requiring speed and ease of use.
MEDAPRINT FOAM represents a complementary solution within the impression disinfection portfolio, positioned alongside immersion disinfectants to address different workflow needs.
MEDAPRINT FOAM delivers a broad antimicrobial spectrum of disinfection while preserving the dimensional stability, surface detail, and accuracy of dental impressions.
It is fully compatible with commonly used impression materials, including alginates, silicones, polyethers, and hydrocolloids.
Properties
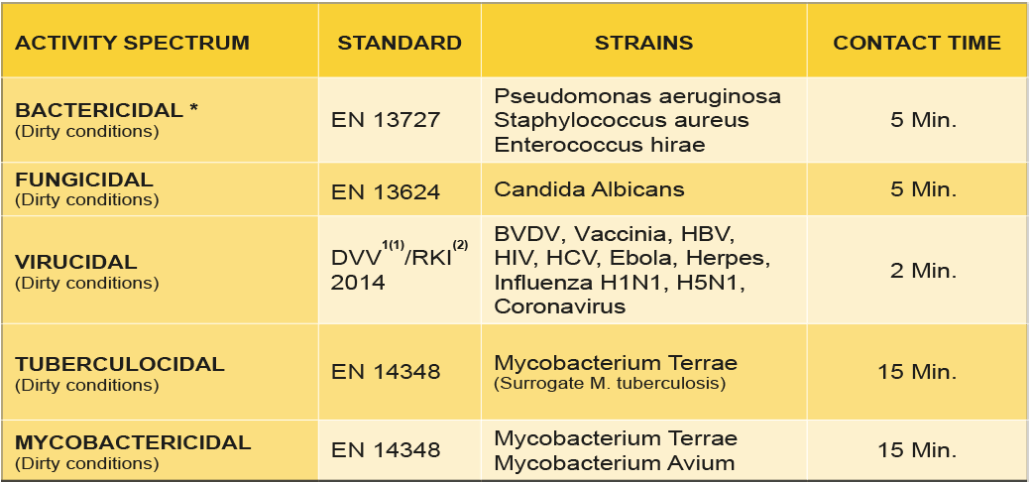
Microbiological efficacy
* Including all antibiotic resistant bacteria such as Escherichia coli, Klebsiella pneumoniae, Streptococcus pneumoniae, etc.)
(1) DVV: Deutsche Vereinigung zur Bekämpfung der Viruskrankheiten / German Association for the Control of Virus Diseases
(2) RKI: Robert Koch Institute – German Federal Health Authority
Compatibility
MEDAPRINT FOAM is compatible with impressions from alginate, silicone, polyether and hydrocolloids.
MEDAPRINT FOAM is not compatible with disinfecting preparations containing aldehydes.
Packaging
Physical properties
Composition
N- (3-aminopropyl) -N dodecylpropano-1,3-diamine, <5% non-ionic surfactants, corrosion inhibitors, moisturising agent, excipients