NOSOFAST TB is a ready-to-use, foaming, broad-spectrum antimicrobial disinfectant spray, developed to deliver combined cleaning and disinfection of non-invasive medical device surfaces.
It provides reliable antimicrobial performance to support the protection of patients and healthcare personnel as part of routine infection prevention and control practices.
The alcohol-free formulation is optimized for use on sensitive medical equipment surfaces, including incubators, patient monitors, plexiglass, and other delicate materials where alcohol-based products may be unsuitable.
In addition, NOSOFAST TB is indicated for routine disinfection of general medical device surfaces such as beds, stretchers, work benches, and related healthcare equipment.
NOSOFAST TB is residue-free after drying, odorless, and formulated to provide high material compatibility, enabling frequent application without adverse effects on treated surfaces.
Properties
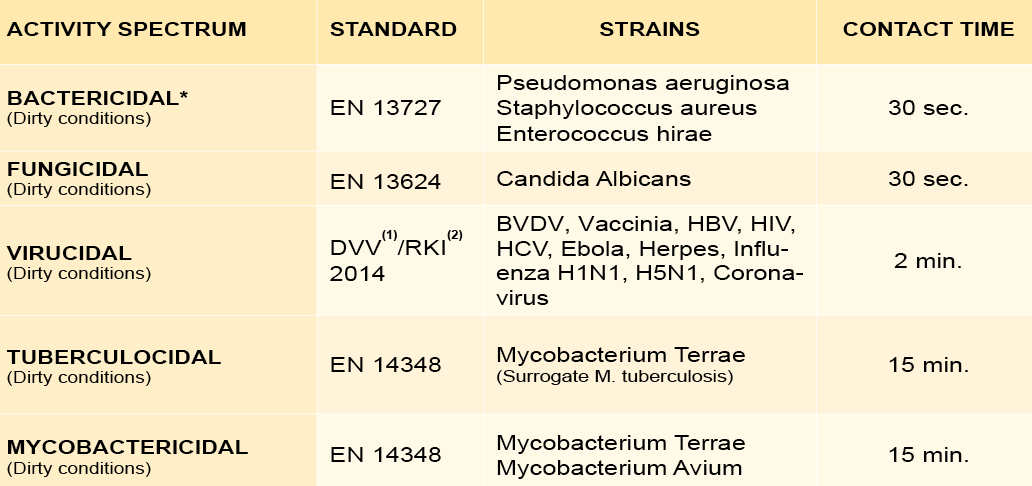
Microbiological efficacy
* Including all antibiotic resistant bacteria such as MRSA, Escherichia coli, Klebsiella
pneumoniae, Streptococcus pneumoniae, etc.)
(1) DVV: Deutsche Vereinigung zur Bekämpfung der Viruskrankheiten (German
Association for the Control of Virus Diseases)
(2) RKI: Robert Koch Institute – German Federal Health Authority
Packaging
Physical properties
Composition
N-(3-aminopropyl)-N-dodecylpropano-1,3-diamine, non-ionic surfactants <5%, corrosion inhibitor, wetting agent, excipients